Environment & Energy
Related: About this forumImproved efficiency of selective photoionization of palladium isotopes via autoionizing Rydberg states.
The paper I'll discuss in this post is this one: Locke, C.R., Kobayashi, T. & Midorikawa, K. Improved efficiency of selective photoionization of palladium isotopes via autoionizing Rydberg states. Appl. Phys. B 123, 33 (2017).
It may seem a little odd to post about this paper, with its admittedly esoteric title, in the E&E forum, but the reason for doing so should be clear enough with a little elaboration, since it it is very much involved with an irrational fear that has left the world in flames, and has caused vast destruction of the natural environment in service to the disastrous idea that so called "renewable energy" is somehow not only sustainable, which it isn't, but that the reactionary impulse to build it is somehow desirable and preferable to far more sustainable nuclear energy.
We have here members of the "renewable energy will save us" cults who make the absurdly specious claim, an example of transparent circular reasoning, that so called "renewable energy" is acceptable because it exists. To my mind, this is equivalent to stating that having pancreatic cancer is a good idea because pancreatic cancer exists. And let's be clear, it is a cancerous notion that to save the world we have industrialize vast stretches of wilderness, as is increasingly clear, on a continental scale, to be "green." It is Orwellian double speak to claim that running strips of asphalt through forests to service microplastic spewing wind turbines, for just one example, is "green." Chain sawing trees or, in deserts, centuries old Joshua trees - as recently showed up in the media in connection with a solar industrial plant in the Mohave desert - is destroying green things.
How this isn't obvious, escapes me.
Antinukism, a kind of modern cargo cult, is an intellectual and environmental cancer, inasmuch it tolerates and encourages the use of fossil fuels. Antinukism is the sole reason for the reactionary enthusiasm of so called "renewable energy," dependence on which was abandoned in the late 19th and early 20th century for a reason. Driving this horror of antinuke mysticism, is the notion that springs from the widely held nonsensical idea that if something is radioactive it must be feared and isolated in such a way that no one ever, anywhere, at anytime should ever even being allowed, even conceivably be exposed to it. This is weird and ignorant on inspection, since potassium is an essential element, without which life is impossible, and all potassium is radioactive. It follows that exposure to radiation is essential to life, although one may reasonably and should argue that the "dose makes the poison." The question is not whether radioactivity is "safe" but rather how much is required - a function of the need for potassium - and, beyond, how much is tolerable. There are obviously points where exposure to radiation is harmful; but these points, should be involved with comparative risk: It is not true that the presence of trace amounts of the cesium isotope 137Cs in German cows in the year after the explosion of the famous Chernobyl reactor justified the release of vast quantities of cancerous and far more cancerous polycyclic hydrocarbons from coal plants, not to mention hundreds of millions of tons of carbon dioxide.
How radioactive a material is inversely proportional to its lifetime. The longer the half-life of a radioactive material is, the less radioactivity it exhibits. The low radioactivity of potassium, owing to the presence of one of its naturally occurring isotopes, 40K is a function of its very long half-life, over 1.2 billion years, allowing it to persist, like uranium and thorium, since its formation in the star or stars in which all of the elements on our radioactive planet Earth, with the exception of hydrogen, lithium, beryllium and boron, extrastellar elements. (Helium on Earth, but not in space, originates from the nuclear α decay of uranium, thorium and their daughters, and originates in rock.)
This brings me to to the point of the paper that titles this post.
From the introductory text:
I take issue with the claim that I have placed in bold.
How so?
It is useful to consider the nuclear decay properties of 107Pd , which can be found here: BNL Nuclear Database 107 Pd Here we see the following data:
Radioactive Decay Data
Half life: 2.0512E+14 ( 9.4673E+12) s, or 6.50E+06 a
Ebeta: 9.8700E+03 ( 9.8000E+02) eV
Egamma: 0.0000E+00 ( -1.4505E-12) eV
Ealpha: 0.0000E+00 ( 0.0000E+00) eV
The link may look like gobbledygook to those unfamiliar with this sort of thing but what is found is that the half-life of 107Pd is 6.5 million years, and that the decay energy is connected with an emitted β particle having an energy of 9.87 keV. Most importantly, there is no γ radiation emitted by the isotope. (Egamma zero)
The half-life of 107Pd is also given in seconds at the link as 9.4673 X 1012 seconds, resulting in a decay constant (ln(2)/9.4673 X 1012) of 7.3215 X 10-14 sec-1. When this activity multiplied by Avogadro’s number yields a molar specific activity of 4.4091 X 1010 Beq mol-1, or 4.12431E+08 Beq-g-1. We are told, in the paper that 17% of the Pd obtained from used nuclear fuel is 107Pd with the balance being stable isotopes of palladium. Since this isotope does not release gamma radiation, but is a pure β emitter, with β rays being stopped by a thin layer of metal, we can expect bulk Pd to be self-shielding. Only those atoms near the surface of bulk palladium will emit radiation to the immediately surrounding air.
A commonly used popular unit for radioactivity is the Curie (Ci) which is equal (exactly) to 3.7 X 1010 Beq (decays sec-1). This implies that the radioactivity of palladium isolated from used nuclear fuel under the conditions described in the paper – the 17% figure may vary based on the amount of time the fuel is working in the reactor, the power of the reactor and other things – will be 1.9 microcuries per gram. This is a relatively low level of radioactivity, on a scale of handling well within the levels found, for instance, in a medical imaging facility. In an industrial device containing palladium, perhaps in a chemical reactor, it's radioactivity would represent low risk when used by trained technicians aware of its mild radioactivity.
The distribution of isotopes in fissiogenic palladium will differ from that from "natural palladium" formed in the stars whose explosion led to all of Earth's elements. The paper reports this distribution as follows, again, subject to the conditions of a particular nuclear reactor and its fuel type:
It is important to note that only two of the mass numbers are odd, 105 and 107, which is important in this context.
However useful 107Pd might be - I contend that its radioactivity, coupled with its chemical catalytic properties make it uniquely useful - right now, the main use of palladium is in automotive exhaust catalysts, along with another even more rare element also found in used nuclear fuel, rhodium, the latter for the destruction of problematic NOx pollutants. It is believed, probably overkill in my view as 107Pd is a pure β emitter, but surely not in the case very many other people, that the use of 107Pd in automotive catalysts would be "too dangerous." Therefore there is a motivation for separating it from the other stable palladium isotopes found in used nuclear fuel.
A "Rydberg state" is an excited state of an atom, described by quantum mechanics, in which an electron has been promoted to a higher energy level determined by its principle quantum number, an orbital, generally a very high level. Because electrons in this these states are unpaired, this state has magnetic properties. Atomic nuclei also can have magnetic properties: They can be bosons, in which the magnetic moment results from an integer spin, including zero in which case there is no spin at all, or be fermions, in which case they have half integer spin.
These magnetic properties are very useful in the analytical technique NMR, nuclear magnetic resonance, also known in medical use as MRI, magnetic resonance imagining, where the word "nuclear" has been removed to avoid scaring idiots. (MRI does not involve radioactivity in most cases.) In general, isotopes with an odd atomic mass number will be fermions (which always have a nuclear spin) and even numbered nuclei will be bosons which may or may not have a nuclear spin.
On a somewhat simplistic level, the coupling of the magnetic electron in a Rydberg State with that of a nucleus having a nuclear spin will confer properties that will allow for the separation of isotopes using very finely tuned lasers, since the monochromatic nature of laser light can differentiate the energy transitions associated with electrons. This will allow selective ionization based on mass number. A graphic in the paper shows how these transitions differ between odd and even isotopes:
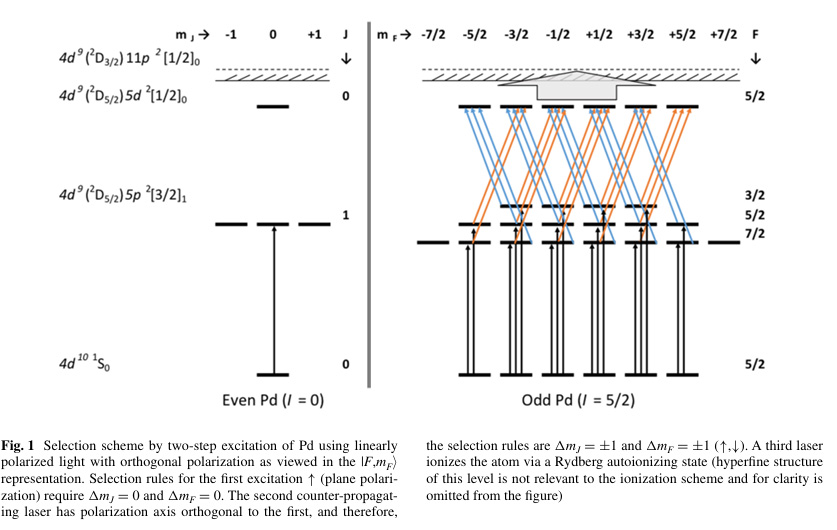
Atoms that are ionized via Rydberg states will have electric charge and can thus be separated in electric fields.
In this system - there may not be a point in this space for belaboring the concept with too much more detail, which in any case is arcane - one would have non-radioactive palladium with all even numbered mass numbers, and a mixture of radioactive 107Pd and non radioactive 105Pd.
β radiation is intermediate in range between α radiation, which is not penetrating at all and γ radiation which requires significant shielding to attenuate it, usually using heavy atoms like lead, uranium or thorium. While β radiation can be stopped with a very thin layer of metal, it can penetrate in significant levels in air.
I am working to convince my son to consider Brayton cycle nuclear reactors in which, as is the case in the most widely used Brayton devices in industrial use, jet engines, the working fluid is neither helium nor carbon dioxide, as in currently developed high temperature gas cooled reactors, but rather air. The drawback of this approach would be the formation, at high temperatures and pressures, of NOx, which palladium catalysts can destroy. An advantage of a radioactive palladium catalyst in such a system would be to exploit the value of high energy radiation for the destruction of CFC's, HFC's and N2O, similar to the process that takes place in the upper atmosphere, leading in the case of CFC's and N2O to ozone depletion. (N2O is a side product of agriculture, and cannot be eliminated from use without inducing vast worldwide starvation.)
To my mind there is a thus a use for the odd numbered isotopes of palladium, where 107Pd would be superior to natural palladium owing to its radioactivity.
The separation of 107Pd will not be absolutely perfect. A nice Japanese paper considers "acceptable" levels of 107Pd for general use while going over the types of use of palladium in current settings:
Tomoyuki Takahashi, Kayoko Iwata, Sota Tanaka, Naoki Takashima, Tomoyuki Ikawa & Sentaro Takahashi (2018) Lifecycle of palladium in Japan: for setting clearance levels of 107Pd, Journal of Nuclear Science and Technology, 55
There are a plethora of papers on this subject, including papers relating to the recovery of other very valuable elements in used nuclear fuel, which antinukes and "I'm not an antinuke" antinukes call, in their tiresome ignorance, "nuclear waste."
Another recent paper covering the many other valuable elements that can be isolated from used nuclear fuel in general is this one:
Serban Nicolae Stamatin, Walter Bonani, Darina Blagoeva, Marco Cologna, Waste to resource: a review of critical raw materials recovery from spent nuclear fuel, Sustainable Energy Technologies and Assessments, Volume 88, 2026, 104926
This paper gives the percentage of industrial demand for high value elements in Europe (which lacks geological resources for many of them) that can be obtained from used nuclear fuel. Notably, used nuclear fuel could pretty much meet nearly all of the European demand for ruthenium.
I wish you a pleasant Sunday.
Orrex
(67,188 posts)And although much of the detail elude my shriveled little brain, it's still a fascinating read.
Thanks!